- Home
-
Industries
-
Environment Testing
- Construction Dust Testing
- Drinking Water Testing
- Emerging Contaminants Testing
- Sea Water Testing
- Sludge Testing
- Soil Testing
- Solid Waste
- Stack Emissions Testing
- Stormwater Testing
- Subsurface Vapour Intrusion Assessment
- Surface Water Testing
- Wastewater Testing
- Watersheds and Rivers Testing
- Well Water Testing
- Asbestos Testing
- Mold Testing
-
Food Testing
- Beverage Testing
- Biscuit Testing
- Candy Testing
- Canned Food Testing
- Coffee Testing
- Condiments Testing
- Cooking Oil Testing
- Fast Food and Snack Testing
- Infant Food Testing
- Liquor Testing
- Meat and Meat Product Testing
- Milk & Dairy Testing
- Pet Food Testing
- Refrigerated and Frozen Food Testing
- Seafood Testing
- Tomato Products Testing
- Agricultural Products & Crops Testing
- Material Testing
- Chemical Products Testing
- Petroleum Products Testing
- Personal Care & Beauty Products Testing
- Household and Apparel Products Testing
- Healthcare Products Testing
- Building Products Testing
- Stationery and Office Supplies Testing
- Safety Testing of Nano Products
- Children Products Testing
-
Environment Testing
-
Services
- Agriculture & Crop Analytical Services
- Energy Analytical Services
- Environmental Analytical Services
- Food Analytical Services
- Material Characterization Services
-
Pharmaceutical Analytical Services
- Biopharmaceutical Characterization Services
- Deformulation (Reverse Engineering) Analysis Services
- Pharmaceutical and Medical Device Failure Analysis Services
- Pharmaceutical Impurity Testing Services
- Pharmaceutical Separation and Purification Services
- Preformulation Analysis Services
- Stability Analysis Services
- Compendial Testing
- Dissolution Testing
- Water Content Determination
- Pharmaceutical Water Testing
- Potency Testing
- Pharmaceutical Microbiology Testing
- Retail Products Analytical Services
- Textile Testing Services
- Karl Fischer (KF) Moisture Testing
-
Recommended Services
- Failure Analysis and Investigations
- Food Testing
- Pharmaceutical Testing
- Surfactant Testing
- Cosmetics and Skin Care Products Testing
- Karl Fischer (KF) Moisture Testing Service
- Textile Testing
- Microplastic Analysis and Testing
- Mold Testing Service
- Lubricating Oil Analysis and Testing
- Alloy Material Testing
- Deformulation Service
- Semiconductor Testing Services
- Techniques
- AI SmartQC Platform
-
Resources
- Regulatory Resources
- Blog
- Application Notes
- Video Library
- White Paper
- Flyer
- Case Study
- Fee Schedule
-
Protocol
- Protocol for Deformulation of Pharmaceutical Products
- Experimental Procedure for Determining Trace Moisture in Food by Karl Fischer Method
- Experimental Procedure for Determining Moisture Content in Interior Wall Paint Using Karl Fischer Method
- Testing of Color Fastness to Rubbing for Textiles
- Qualitative Identification of Textile Fibers by Five Experimental Methods
- Tablet Dissolution Test - Determination of Dissolution Rate and Dissolution Rate of Azithromycin Dispersible Tablets
- Determination of Chemical Oxygen Demand (COD) in Water - Potassium Dichromate Method
- How to Detect Bacteria in Food: Total Bacterial Count Procedure
- Testing Coliforms in Food: MPN & Plate Count Methods
- Determination of Aflatoxin B1 in Food: ELISA & TLC Methods for Accurate Detection
- Determination of Molds and Yeasts in Food: Plate Count Methods for Accurate Detection
- Determination of Lubricating Oil Liquid Density - Density Meter Method
- Determination of Acid Value in Lubricating Oil - Potentiometric Titration Method
- ELISA Experimental Procedure for Detecting Aflatoxin B1 in Peanuts
- Experimental Guide: Standard Procedures for Semiconductor Device DC Parametric Testing
- Failure Analysis Protocol: Locating Chip Leakage Points Using EMMI and OBIRCH
- Careers
- About
- Contact
- Home
- Services
- Retail Products Analytical Services
- Failure Analysis Services
- Electroplating Problems Analysis
Electroplating Problems Analysis
InquiryThrough our global network of testing experts and analytical equipment including chromatography (HPLC, GC, GC/MS) and atomic absorption spectroscopy (AAS, GFA, FIAS), Our goal is to provide test services as efficiently as possible to maximize our customers' profits. For more information about our services, contact one of our experts today.
Note: this service is for Research Use Only and Not intended for clinical use.
Electroplating is a process that uses electric current to reduce dissolved metal cations so that they can form a thin coherent metal coating on an electrode. By using this method, the surface properties of a product or material(such as abrasion resistance, corrosion protection and lubricity)can be modified to meet different physical and chemical demands. For example, chromium plating is plated on many objects such as car parts, bath taps, kitchen gas burners, wheel rims to prolong the life of these objects due to the fact that chromium is corrosion-resistant. Electroplating is also used in making expensive jewelry, like 14K or 18K gold-plated jewelry. Copper is another popular electroplating option because it is abundant in nature and relatively inexpensive. And due to its natural electrical conductivity, copper is usually chosen as the metal for the manufacture of circuit boards and other electronic components. It also provides excellent protection against corrosion, since it is less hazardous to the environment than many other plated metals.
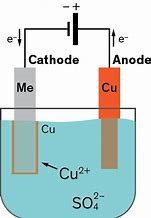 Fig.1 A picture of electroplating process.
Fig.1 A picture of electroplating process. Fig.2 Examples of electroplating application.
Fig.2 Examples of electroplating application.
Electroplating can change the conductivity, hardness, resistance, brittleness, luster and appearance of objects. It is significant to select suitable coating material to improve the service life and performance of products. If the electroplated coating on the surface of the metal products fails, the internal material may be easily corroded when exposed to the environment and further causes the material to fail. Every year, a large number of materials will be invalidated due to the failure of the electroplated coating.
Sharp edges, inside corners, deep narrow grooves, scratches and cracks, threads, inside diameters differences are all the factors that can cause electroplating problems. Poor adhesion is another major problem that can negatively affect the performance and longevity of an electroplated coating, resulting in the flaking of electroplated coating and failure of materials or products. Many factors can cause the flaking of electroplated coating, such as inadequate surface activation, excessive coating thickness and current interruptions during electroplating. Many tests can be performed to indicate the presence of flaking in a plated part, such as bend test and heat-quench test.
We offer professional testing for your electroplating characterizations in the following industries:
Automobile
Shipbuilding industry
Civil engineering
Aerospace
Electronic and electrical industries
Jewelry
Daily necessities
Other related products or materials
Testing laboratories of Alfa Chemistry possess many professional equipment for ISO and ASTM method testing. Here are some examples:
Scanning Electron Microscopy-Energy Dispersive Spectrometer (SEM-EDS)
Transmission Electron Microscope (TEM)
Fourier Transform Infrared Spectroscopy (FTIR)
Differential Scanning Calorimeter (DSC)
Thermal Gravity Analysis (TGA)
Universal Testing Machine (UTM)
X-Ray Fluorescence (XRF)
X-Ray Diffraction
Alfa Chemistry is professional in solving electroplating problems for many industries. Our research and testing services can provide you with accurate and reliable tests results within the time constraints. Please feel free to contact us if you have any concerns or would like us to perform any electroplating testing. Check our instruments list to see our capabilities.
- Recommendation
- Ordering Process
- Ask a Question
Do not know how to place an order, please refer to the flow chart shown below.
Submit quotation request |
A technical manager will contact you within 24 hours |
You will review and approve the final price and place an order |
Confirm with you and make the payment |
Instruct you to ship your samples and form |
Analytic report delivery |

